The natural world presents us with an extraordinary palette of colored minerals and crystals, from deep purples and vibrant greens to striking pinks and reds. Among these, pink-hued rocks and crystals have captivated collectors, jewelers, and geology enthusiasts for centuries. But what creates these beautiful colors? The answer lies in the fascinating intersection of chemistry, physics, and geology—specifically, in the trace elements embedded within crystal structures.
This comprehensive guide explores the scientific principles behind crystal coloration, with a particular focus on pink minerals. Whether you’re a mineral collector, jewelry enthusiast, or simply curious about the natural world, understanding the chemistry of color will deepen your appreciation for these geological wonders.

The Fundamental Science of Crystal Color
Crystals are remarkable natural structures where atoms, ions, or molecules are arranged in repeating three-dimensional patterns. This ordered arrangement is what gives crystals their distinctive shapes and many of their physical properties, including color. Understanding what makes a crystal appear pink, blue, or any other color requires examining several interconnected factors.
Primary Factors Influencing Crystal Color
The color you observe in any mineral or crystal results from complex interactions between light and matter. Several key factors determine the final appearance:
- Mineral composition: The primary chemical elements and compounds that form the crystal’s base structure
- Trace element incorporation: Small amounts of “impurity” elements that substitute into the crystal lattice
- Crystal structure and symmetry: How atoms are spatially arranged affects how they interact with light
- Light absorption and reflection: Which wavelengths of light are absorbed versus reflected determines perceived color
- Formation conditions: Temperature, pressure, and chemical environment during crystal growth
- Physical defects: Structural imperfections can create color centers that absorb specific wavelengths
When white light—which contains all visible wavelengths—strikes a crystal, certain wavelengths are absorbed by the material’s atomic structure. The wavelengths that aren’t absorbed are reflected back to our eyes, and these reflected wavelengths determine the color we perceive.
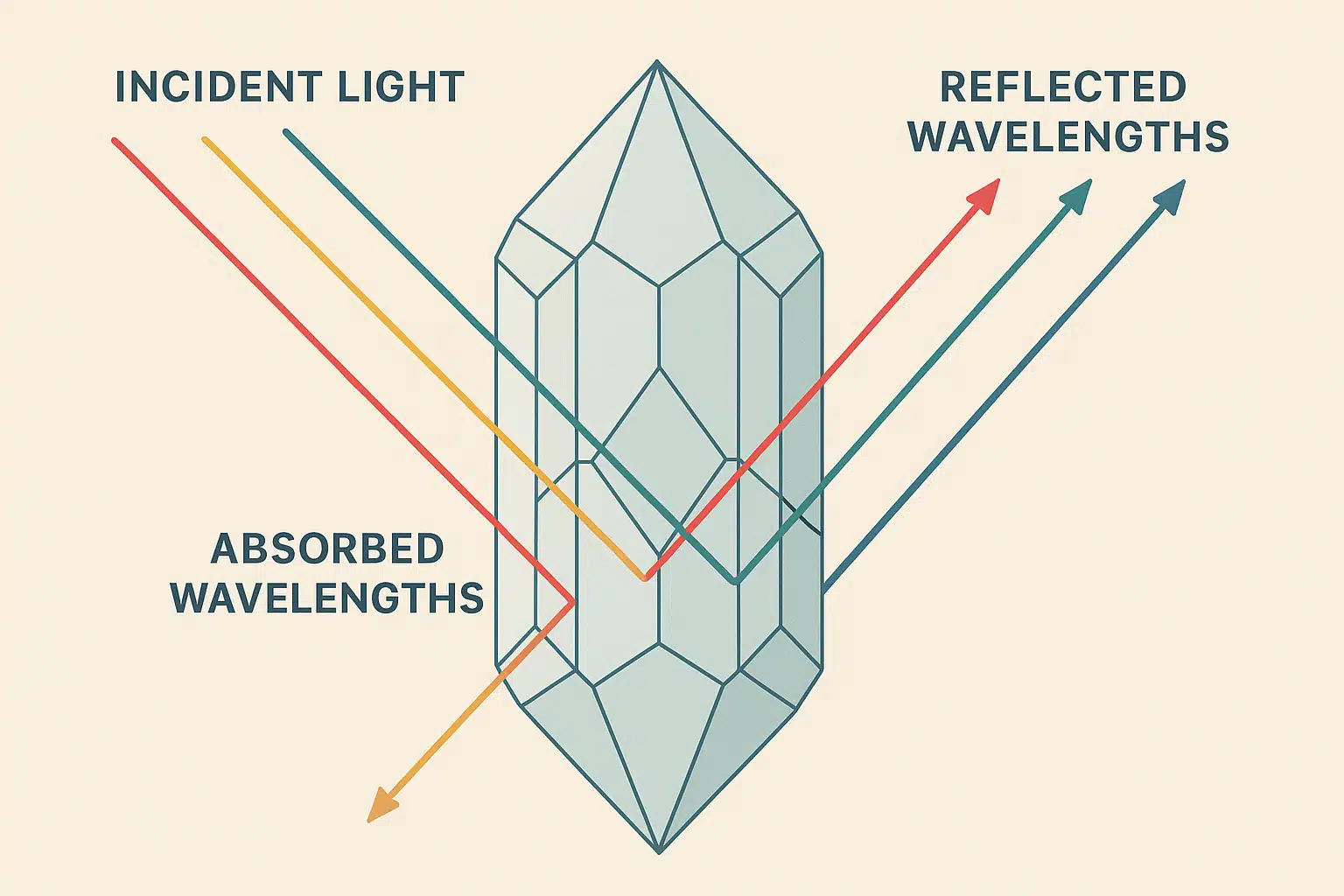
Trace Elements: The Color Creators
While the base mineral composition establishes the crystal’s fundamental properties, trace elements—present in concentrations as small as parts per million—often have a disproportionate effect on color. These elements work as chromophores, substances that cause color by absorbing particular wavelengths of light.
Manganese: The Pink Producer
Manganese is one of the most common trace elements responsible for pink coloration in minerals. When manganese ions substitute for other elements in a crystal structure, they create electronic transitions that selectively absorb green and yellow wavelengths of light. The remaining reflected light appears pink to our eyes.
The intensity of pink coloration depends on several variables:
- Concentration: Higher manganese content generally produces deeper, more saturated pink hues
- Oxidation state: Manganese can exist in different ionic forms (Mn²⁺, Mn³⁺, etc.), each producing slightly different colors
- Crystal environment: The surrounding atoms and the specific crystal structure influence how manganese ions interact with light
- Co-occurring elements: Other trace elements present can modify or enhance the pink coloration
Iron: The Versatile Color Agent
Iron is perhaps the most versatile color-producing element in the mineral world. Depending on its oxidation state and concentration, iron can create colors ranging from pale yellow and green to deep red, brown, and even blue-green hues.
In the context of pink minerals, iron plays several roles:
- Ferrous iron (Fe²⁺): Typically produces green to blue-green colors but can contribute to pink when combined with other chromophores
- Ferric iron (Fe³⁺): Creates warm tones including yellow, orange, red, and can enhance pink coloration
- Iron-manganese interaction: When both elements are present, they can work synergistically to produce unique pink shades
- Charge transfer mechanisms: Movement of electrons between iron ions and other elements can create distinctive colors

Notable Pink Minerals and Their Chemistry
Examining specific examples helps illustrate how chemical composition translates into the beautiful pink minerals found in nature and used in various applications.
Rose Quartz: The Classic Pink Crystal
Rose quartz is one of the most recognizable and beloved pink minerals. This variety of quartz (silicon dioxide) typically displays a delicate pink to rose-red color. For many years, scientists debated the exact cause of its coloration, but research has revealed a complex picture.
The pink color in rose quartz can result from:
- Microscopic fiber inclusions: Tiny fibers of a mineral called dumortierite create a pink appearance through light scattering
- Trace amounts of titanium, iron, and manganese: These elements working together produce the characteristic color
- Radiation-induced color centers: Natural radiation exposure can create structural defects that contribute to coloration
Rose quartz is widely used in jewelry, decorative objects, and various wellness applications. Its relatively abundant availability and appealing color make it popular worldwide.
Pink Tourmaline: Complex Chemistry Creates Stunning Gems
Tourmaline represents one of the most chemically complex mineral groups, with a general formula that can accommodate dozens of different elements. This chemical flexibility results in tourmaline occurring in virtually every color imaginable, including spectacular pinks.
Pink tourmaline, sometimes called rubellite when particularly vivid, owes its color to:
- Manganese content: The primary chromophore in most pink tourmalines
- Lithium presence: Often found alongside manganese in pink varieties
- Iron and manganese balance: The ratio between these elements affects whether the stone appears pink, red, or brownish
- Crystal orientation: Tourmaline is pleochroic, meaning it shows different colors when viewed from different angles
Pink tourmaline is highly valued in fine jewelry and commands premium prices, especially for stones with vivid, saturated color and good clarity.
Rhodochrosite: Manganese’s Masterpiece
Rhodochrosite is a manganese carbonate mineral that displays some of the most intense and beautiful pink to red colors in the mineral kingdom. Unlike minerals where manganese is merely a trace element, rhodochrosite’s base composition is manganese carbonate (MnCO₃), making manganese an essential component rather than an accessory.
The color intensity in rhodochrosite varies based on:
- Manganese purity: The highest quality specimens with minimal substitution show the deepest colors
- Calcium content: Calcium can replace some manganese, generally lightening the color
- Iron substitution: Iron replacing manganese tends to shift the color toward brownish tones
- Crystal formation patterns: Banded specimens show color variations reflecting changing chemical conditions during growth
Morganite: Beryl’s Pink Variety
Morganite is the pink to peachy-pink variety of beryl, the same mineral species that includes emerald and aquamarine. This gemstone demonstrates how trace elements can dramatically transform a mineral’s appearance—pure beryl is colorless, but small amounts of manganese create morganite’s delicate colors.
Key characteristics include:
- Manganese as chromophore: Mn³⁺ ions are responsible for the pink coloration
- Color stability: Morganite’s color is generally stable and doesn’t fade with exposure to light
- Heat treatment: Some morganite is heat-treated to remove yellow or orange tones, enhancing the pink color
- Excellent clarity: Morganite typically forms with good transparency, making it ideal for faceted gemstones

Common Misconceptions About Crystal Color
Despite widespread interest in minerals and crystals, several misconceptions persist about what causes their colors. Clarifying these misunderstandings helps develop a more accurate appreciation of mineralogy.
Misconception: Each Mineral Has One Specific Color
Many people assume that minerals can be reliably identified by color alone. In reality, most minerals can occur in multiple colors depending on trace element content, structural defects, and formation conditions. Quartz, for example, can be colorless (rock crystal), purple (amethyst), yellow (citrine), pink (rose quartz), brown (smoky quartz), or even black.
This variability means that color, while useful, should never be the sole identification criterion. Other properties like crystal shape, hardness, cleavage, and density are more reliable diagnostic features.
Misconception: All Pink Minerals Contain the Same Coloring Agent
While manganese is a common cause of pink coloration, it’s not the only mechanism. Different minerals achieve pink colors through various chemical and physical processes:
- Cobalt: Creates pink in some minerals like erythrite (cobalt arsenate)
- Color centers: Structural defects caused by radiation can produce pink without metal ion chromophores
- Inclusion-based color: Microscopic mineral inclusions can create apparent pink coloration through light scattering
- Multiple element combinations: Complex interactions between several trace elements can yield pink hues
Misconception: Color Is Purely About Chemical Composition
While chemistry plays the dominant role, physical factors also significantly influence color. The conditions under which a crystal forms—including temperature, pressure, cooling rate, and exposure to radiation—can affect the final color even when chemical composition remains constant.
Additionally, some minerals exhibit phenomena like:
- Pleochroism: Different colors when viewed from different directions
- Chatoyancy: A cat’s-eye effect from parallel fiber inclusions
- Asterism: Star-like reflection patterns from intersecting inclusions
- Color change: Different appearance under various light sources
Misconception: Deeper Color Always Means Higher Quality
In gemstones, color intensity is just one quality factor among many. While vivid, saturated colors are often desirable, they must be balanced with other characteristics like clarity, cut quality, and overall appearance. An overly dark stone may appear nearly opaque, while too pale a color may lack visual impact. The most valuable gemstones typically show optimal color saturation—vivid but not too dark.

Treatments and Enhancements
The gem and mineral industry commonly employs various treatments to enhance or modify colors. Understanding these practices helps consumers make informed decisions when purchasing pink crystals and gemstones.
Heat Treatment
Controlled heating is one of the oldest and most widely accepted gemstone treatments. Heat can:
- Enhance existing color: Make pale pink more vivid
- Remove unwanted tones: Eliminate brown or yellow secondary colors
- Create color: Transform colorless material into colored varieties
- Improve clarity: Dissolve or modify certain types of inclusions
Heat treatment is generally permanent and stable. Many heat-treated gemstones are indistinguishable from naturally colored ones without sophisticated testing. In the gemstone trade, heat treatment is so common for certain stones that untreated specimens may command premium prices.
Irradiation
Exposure to various types of radiation can induce or modify color in some minerals. This process creates color centers—structural defects that absorb specific wavelengths of light. Irradiation treatment must be carefully controlled to ensure the resulting material is safe and that the color is stable.
Coating and Dyeing
Some pink minerals and crystals receive surface treatments or are dyed to enhance or create color. These treatments are generally less permanent and should be disclosed by reputable sellers. Surface coatings can wear away with time or cleaning, and dyes may fade with light exposure.
When purchasing crystals or gemstones, always ask about treatments. Reputable dealers will disclose any enhancements, allowing you to make an informed choice.
Practical Applications of Pink Crystals and Minerals
Beyond their scientific interest, pink crystals and minerals serve numerous practical and decorative purposes in modern life.
Fine Jewelry and Adornment
Pink gemstones have become increasingly popular in jewelry design, offering alternatives to traditional diamond-centered pieces. Their appeal spans various markets:
- Engagement rings: Pink gemstones like morganite and pink sapphire offer distinctive alternatives
- Fashion jewelry: Rose quartz and pink tourmaline appear in accessible jewelry pieces
- High-end collector pieces: Rare, exceptional pink gemstones command premium prices
- Custom designs: Unique colors allow for personalized, meaningful jewelry
Mineral Collecting and Display
Mineral collecting is a worldwide hobby with enthusiasts ranging from casual collectors to serious specialists. Pink minerals are particularly sought after for their aesthetic appeal and the scientific principles they illustrate. Collectors may focus on specific mineral species, geographic sources, or crystal formations.
Decorative and Interior Design Uses
Polished pink minerals serve as decorative elements in homes and offices. Large specimens, bookends, spheres, and carved items bring natural beauty into interior spaces. The soft, calming hues of pink minerals complement various design aesthetics from minimalist modern to eclectic bohemian.
Educational and Research Purposes
Pink minerals serve as excellent teaching tools for earth science education, demonstrating concepts in chemistry, crystallography, and geology. Educational mineral collections often include pink varieties to illustrate trace element effects on color.

Identifying and Evaluating Pink Minerals
For collectors and consumers, developing skills to identify and evaluate pink minerals ensures authentic purchases and helps build knowledge.
Basic Identification Techniques
While definitive mineral identification often requires laboratory testing, several accessible methods provide useful information:
- Visual examination: Observe crystal form, luster, transparency, and color distribution
- Hardness testing: Use a mineral hardness kit to determine position on the Mohs scale
- Specific gravity: Measure density using simple equipment
- Streak test: Examine the color of powdered mineral on unglazed porcelain
- Reaction to light: Observe under different lighting conditions and with UV light
Quality Factors in Pink Gemstones
When evaluating pink gemstones for purchase, consider these quality factors:
- Color: Hue (the primary color), tone (lightness or darkness), and saturation (intensity or purity)
- Clarity: Presence and visibility of inclusions or internal characteristics
- Cut: Proportions, symmetry, and polish quality that affect brilliance and appearance
- Carat weight: Size, with larger stones often commanding disproportionately higher prices
- Treatment status: Whether the stone is natural, enhanced, or synthetic
- Origin: Some geographic sources command premium prices
Avoiding Synthetic and Imitation Materials
The market includes natural minerals, laboratory-created synthetics (same chemical and physical properties as natural), and imitations (different composition but similar appearance). All have legitimate uses, but proper disclosure is essential.
Tips for ensuring authenticity:
- Purchase from reputable sources: Established dealers with good reputations
- Request documentation: Certificates from gemological laboratories for significant purchases
- Learn common imitations: Understand what materials might be substituted
- Be wary of too-perfect specimens: Natural materials typically have some imperfections
- Compare prices: If something seems too inexpensive, investigate further
Caring for Pink Crystals and Gemstones
Proper care ensures that pink minerals and gemstone jewelry maintain their beauty for years or even generations.
Cleaning Guidelines
Different minerals require different care approaches based on their hardness, structure, and any treatments they’ve received:
- Hard, durable gemstones: Can typically be cleaned with warm water, mild soap, and a soft brush
- Softer materials: Require gentler cleaning with just water and a soft cloth
- Treated stones: May need special care to preserve treatments
- Jewelry settings: Consider the metal and setting style when cleaning
Avoid ultrasonic and steam cleaners unless you’re certain they’re safe for your specific material, as these can damage some gemstones and loosen settings.
Storage and Protection
Proper storage prevents damage from scratching, chipping, or environmental factors:
- Separate storage: Keep harder materials away from softer ones to prevent scratching
- Individual compartments: Use jewelry boxes with dividers or soft pouches
- Moderate conditions: Avoid extreme temperature changes and very low humidity
- Light protection: Some materials may fade with prolonged light exposure
- Secure settings: Regularly check jewelry settings to ensure stones remain secure
When to Seek Professional Care
Certain situations require professional jeweler or gemologist attention:
- Setting repairs: Loose stones or damaged prongs
- Professional cleaning: For valuable pieces or when home cleaning is insufficient
- Repolishing: To restore surface luster on scratched gemstones
- Appraisals: For insurance purposes or to determine value
- Identification: To verify authenticity or mineral species
The Broader Significance of Understanding Mineral Chemistry
While this guide has focused on the appealing topic of pink minerals, the principles discussed extend far beyond aesthetic considerations. Understanding how trace elements affect crystal properties has important implications across multiple fields.
Geological and Environmental Insights
The trace elements that color minerals also provide valuable information about the conditions under which rocks formed. Geologists use this information to understand Earth’s history, including:
- Formation environments: Temperature, pressure, and chemical conditions during crystallization
- Element availability: Which elements were present in the environment
- Alteration history: Subsequent changes that affected the rock
- Economic geology: Locating mineral deposits and understanding ore formation
Materials Science Applications
The principles governing color in natural minerals inform the development of synthetic materials for technology:
- Laser technology: Chromium-doped crystals in ruby lasers
- LED development: Understanding how dopants create specific colors in semiconductors
- Ceramic pigments: Creating stable colors for industrial applications
- Phosphors: Materials that emit light when excited, used in displays and lighting
Cultural and Historical Dimensions
Throughout human history, colored minerals have held cultural significance beyond their physical properties. Pink and red minerals have symbolized love, compassion, and vitality across many cultures. Understanding the science behind these materials enriches our appreciation of how humans have valued and used them throughout history.
Conclusion: The Beautiful Intersection of Chemistry and Nature
The question “Why is this rock pink?” opens a window into the fascinating world of crystal chemistry and mineralogy. The answer involves trace elements like manganese and iron, the specific ways atoms arrange themselves in crystal structures, the interaction between matter and light, and the geological processes that bring these factors together.
Pink minerals demonstrate how even minute amounts of certain elements—sometimes just a few atoms per million—can dramatically transform a material’s appearance. This sensitivity to composition makes minerals valuable not just as beautiful objects but as records of geological processes and as inspiration for technological innovation.
Whether you’re drawn to pink crystals for their aesthetic beauty, their scientific interest, their potential in jewelry design, or simply as collectible natural wonders, understanding the chemistry behind their color deepens the appreciation. The next time you encounter a pink gemstone or mineral specimen, you’ll see not just a pretty object but a story written in atoms—a tangible connection to the fundamental processes that shape the material world around us.
From the delicate blush of rose quartz to the vivid brilliance of fine pink tourmaline, these minerals remind us that nature’s palette is both diverse and scientifically explicable. The chemistry of color in crystals represents one of the many ways that scientific understanding enhances rather than diminishes our wonder at the natural world.

