Understanding Calcium Chloride in Cheese Making
Calcium chloride is an essential yet often misunderstood ingredient in the art and science of cheese making. Whether you’re a home enthusiast exploring dairy crafts or simply curious about how commercial cheese achieves its perfect texture and consistency, understanding this compound can transform your appreciation and success in creating quality cheese. This comprehensive guide explores what calcium chloride is, why it matters in modern cheese production, and how it solves common challenges faced when working with commercially processed milk.

What is Calcium Chloride?
Calcium chloride is a mineral compound consisting of calcium and chlorine that readily dissolves in water, forming a clear solution. In its pure form, it appears as white crystalline flakes or granules. This salt has been recognized as safe for food use by international food safety authorities and has applications across various industries, from food processing to agriculture and beyond.
In the context of cheese making, calcium chloride serves a highly specific and important function. It acts as a coagulation aid, strengthening the protein bonds that transform liquid milk into firm, workable curds. This compound is particularly valuable when working with milk that has undergone heat treatment or pasteurization processes, which are standard practices in commercial dairy production worldwide.
The use of calcium chloride in cheese making is measured and precise. Typically, only small amounts are needed—usually measured in fractions of teaspoons per gallon or liter of milk. Despite these modest quantities, the impact on curd formation and final cheese quality can be substantial.

The Science Behind Pasteurization and Calcium Loss
To understand why calcium chloride has become an important tool in modern cheese making, we must first explore what happens to milk during pasteurization. Pasteurization is a heat treatment process designed to eliminate harmful bacteria and extend shelf life, making milk safer for consumption and distribution. This process has become virtually universal in commercial dairy production across the globe.
However, the heating process involved in pasteurization affects the milk’s chemistry in several ways. One significant change is the alteration of calcium’s availability and distribution within the milk. While the total calcium content may remain relatively unchanged, the calcium that exists in soluble, ionic form—the type most active in curd formation—becomes reduced. Some calcium precipitates out or binds with other compounds during heating, making it less available for the coagulation process.
Additionally, heat treatment affects the casein proteins in milk, which are the primary proteins involved in cheese making. These proteins become slightly destabilized and less responsive to coagulation agents. The natural balance that exists in raw, unheated milk—where calcium ions bridge casein proteins together during curdling—becomes disrupted.
This is where calcium chloride enters the picture as a practical solution. By adding back ionic calcium in a readily available form, cheese makers can restore the milk’s ability to form strong, cohesive curds, compensating for the changes caused by heat treatment.
Why Calcium Chloride is Essential for Quality Cheese
The benefits of using calcium chloride in cheese making extend beyond simply making the process possible with pasteurized milk. This ingredient delivers several tangible improvements to both the cheese-making process and the final product quality.

Enhanced Curd Formation and Structure
When calcium chloride is added to milk before the coagulation process begins, it provides additional calcium ions that strengthen the protein network. This results in curds that are firmer, more resilient, and easier to cut and handle. The curds hold together better during cutting and stirring, reducing the formation of fine particles (known as “fines”) that can be lost in the whey, which directly impacts yield.
Improved Cheese Yield
Stronger curd formation means more efficient moisture retention during the initial stages and less protein loss to the whey. For home cheese makers, this might mean getting noticeably more cheese from the same amount of milk. For commercial operations, even small percentage improvements in yield translate to significant economic benefits. Studies have shown that proper calcium chloride use can improve yields by several percentage points.
Consistent and Predictable Results
One of the frustrations in cheese making, particularly for beginners, is batch-to-batch variability. Different milk sources, seasonal variations, and processing differences all affect how milk behaves during cheese making. Adding calcium chloride helps standardize the coagulation process, making results more predictable and consistent regardless of minor variations in the milk’s starting condition.
Faster and More Complete Coagulation
Calcium chloride can reduce the time needed for curds to form and reach the proper firmness for cutting. This not only makes the process more efficient but also reduces the risk of over-acidification that can occur when coagulation takes too long. The coagulation is also more complete, meaning more of the available protein is incorporated into the curd rather than remaining dissolved in the whey.
How Calcium Chloride Works: The Coagulation Process Explained
Understanding the mechanism by which calcium chloride improves curd formation helps cheese makers use it more effectively and appreciate its role in the larger process.
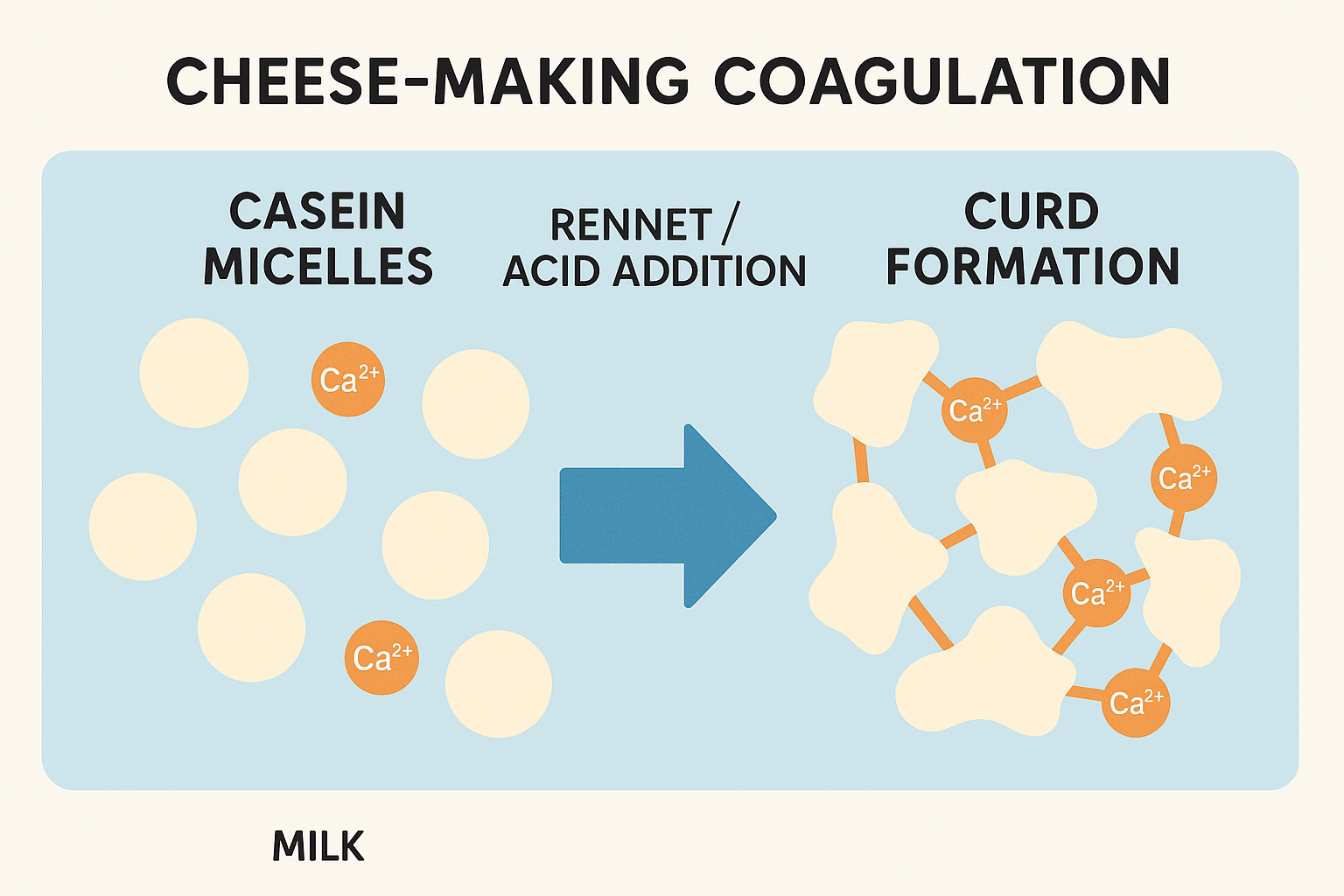
Restoration of Ionic Calcium Levels
When dissolved in milk, calcium chloride dissociates into calcium ions and chloride ions. The calcium ions are the active component, immediately available to participate in the coagulation chemistry. These ions help restore the balance disrupted by pasteurization, bringing the ionic calcium concentration closer to what would naturally exist in raw milk.
Strengthening Protein Bonds
Milk contains several types of proteins, but casein proteins are the stars of cheese making. These proteins exist in milk as complex structures called micelles. During coagulation—whether triggered by acid, enzymes like rennet, or both—these casein micelles must link together to form the three-dimensional network we recognize as curd.
Calcium ions act as bridges between casein molecules, creating strong cross-links that stabilize the developing curd structure. With sufficient calcium present, these bridges form quickly and robustly, resulting in a firm, cohesive curd. Without adequate calcium, the bridges are weaker and fewer, leading to soft, fragile curds that may break apart easily.
pH Buffering and Stability
Calcium chloride also provides some buffering effect in the milk, helping to maintain stability across different acidity levels. This is particularly important in cheeses where acid development plays a significant role. The added calcium helps maintain curd integrity even as pH drops during fermentation, preventing the excessive softening or breakdown that can occur in highly acidic conditions without sufficient calcium.
Practical Application: Using Calcium Chloride in Home Cheese Making
For home cheese makers ready to incorporate calcium chloride into their practice, understanding the proper techniques ensures the best results. Here’s a comprehensive guide to using this ingredient effectively.

Determining the Right Amount
The standard recommendation for calcium chloride use in cheese making is approximately 1/4 teaspoon of calcium chloride solution (at 30-32% concentration) per gallon of milk, or roughly 1/16 teaspoon per liter. For calcium chloride powder or flakes, you’ll need to adjust based on the product’s concentration—always follow the manufacturer’s guidelines.
It’s important not to exceed recommended amounts. Too much calcium chloride can result in curds that are excessively firm and may develop bitter flavors. Starting with the standard amount and adjusting based on your specific milk and desired cheese type is the best approach.
Proper Dilution and Addition Technique
Never add calcium chloride directly to milk in concentrated form. Always dilute it first in a small amount of cool, non-chlorinated water—typically about 60 milliliters (1/4 cup) of water per dose. This dilution ensures even distribution throughout the milk and prevents localized areas of high concentration that could affect curd quality.
Add the diluted calcium chloride solution to your milk before heating or at the beginning of your recipe, well before adding cultures or coagulants. Stir thoroughly to distribute it evenly throughout the milk. The calcium chloride should be fully incorporated before you proceed with the next steps of your cheese-making process.
Step-by-Step: Making Cheese with Calcium Chloride
Here’s a general workflow for incorporating calcium chloride into a basic cheese-making process:
- Prepare Your Calcium Chloride Solution: Measure the appropriate amount of calcium chloride and dilute it in cool water, stirring until completely dissolved.
- Start with Cool Milk: Pour your milk into your cheese-making pot while it’s still cool.
- Add Calcium Chloride: Pour the diluted calcium chloride solution into the milk and stir gently but thoroughly for about one minute to ensure even distribution.
- Heat the Milk: Begin heating your milk to the temperature specified in your recipe, following your normal procedure.
- Add Cultures: Once you reach the appropriate temperature, add your starter cultures as directed by your recipe.
- Add Coagulant: After the culture has had time to ripen the milk (if required by your recipe), add your rennet or other coagulant.
- Allow Coagulation: Let the milk sit undisturbed for the coagulation period. You should notice that the curd forms more firmly and cleanly than without calcium chloride.
- Proceed with Your Recipe: Continue with cutting, cooking, draining, and forming your cheese according to your specific recipe.
Different Cheese Types and Calcium Chloride Requirements
While calcium chloride benefits most cheese varieties made from pasteurized milk, some types benefit more than others, and the application may vary slightly depending on the cheese style.
Fresh and Soft Cheeses
Cheeses like mozzarella, ricotta, paneer, and queso fresco particularly benefit from calcium chloride. These cheeses rely on achieving a good curd set in a relatively short time, and the firmer curds resulting from calcium chloride addition make them much easier to work with. For stretched cheeses like mozzarella, the improved curd structure also contributes to better stretching characteristics.
Hard and Aged Cheeses
Cheddar, gouda, parmesan, and similar aged cheeses also benefit significantly from calcium chloride. The firmer initial curd leads to better moisture control during pressing and ultimately contributes to proper texture development during aging. The improved protein matrix also supports the development of desirable texture and prevents defects during the maturation period.
Acid-Set Cheeses
For cheeses coagulated primarily through acidification rather than enzyme action—such as chèvre or some ricotta styles—calcium chloride still plays an important role. The added calcium helps maintain curd integrity in the acidic environment, preventing excessive softness or syneresis (whey weeping).

Common Misconceptions and Questions About Calcium Chloride
Despite its widespread use in cheese making, several misconceptions about calcium chloride persist. Addressing these helps both beginners and experienced cheese makers use this ingredient with confidence.
“Calcium Chloride is Only for Commercial Operations”
This is false. While commercial cheese makers certainly use calcium chloride, it’s equally valuable for home cheese makers. In fact, home cheese makers often benefit even more because they typically work with retail milk that has been highly pasteurized and may have lower available calcium than the milk used by commercial operations. Calcium chloride is affordable, widely available, and easy to use at home.
“It Changes the Taste of Cheese”
When used at recommended levels, calcium chloride is completely tasteless and does not affect the flavor of the finished cheese. It’s a mineral salt that becomes incorporated into the cheese structure in quantities too small to impart any flavor. The improved texture and consistency it provides actually allow the intended flavors to develop more properly.
“All Milk Needs Calcium Chloride”
Raw, unpasteurized milk typically contains adequate available calcium and may not require calcium chloride addition. However, since most cheese makers worldwide work with pasteurized milk due to availability and safety considerations, calcium chloride becomes a valuable tool. The decision to use it depends on your milk source and the results you’re achieving. If your curds are consistently weak or your yields are lower than expected, calcium chloride is likely to help.
“More is Always Better”
This is definitely not true. Excessive calcium chloride can lead to problems including overly firm, rubbery curds, bitter flavors, and texture defects in the finished cheese. Following recommended dosage guidelines is important. If you’re not getting the results you want with standard amounts, the problem may lie elsewhere in your process rather than requiring more calcium chloride.
“Calcium Chloride is Dangerous or Unhealthy”
Food-grade calcium chloride is recognized as safe by food safety authorities worldwide. It’s a source of dietary calcium, an essential mineral. The amounts used in cheese making are small, and most of it ends up in the whey rather than the finished cheese. It poses no health risks when used appropriately in food applications.
Troubleshooting Common Issues Related to Calcium
Understanding how calcium affects cheese making helps diagnose problems when they arise. Here are common issues that relate to calcium levels and how to address them.
Weak or Fragile Curds
If your curds are soft, break apart easily during cutting, or produce excessive fines, insufficient calcium is a likely cause. This is especially common when using ultra-pasteurized milk or milk that has been stored for extended periods. Solution: Add calcium chloride at the recommended rate or slightly increase if you’re already using some.
Long Coagulation Times
If your milk takes much longer than the recipe indicates to form a clean break, low calcium may be the culprit. Calcium is essential for efficient rennet action, and without it, coagulation is sluggish. Solution: Ensure you’re adding calcium chloride, and verify your rennet is fresh and properly stored.
Poor Cheese Yield
If you’re getting noticeably less cheese than expected from your milk, weak curd formation due to low calcium means you’re losing protein to the whey. Solution: Add calcium chloride to strengthen curd formation and improve protein retention.
Texture Defects During Aging
Some texture problems in aged cheeses, such as excessive softness, short or crumbly texture, or poor body, can trace back to inadequate calcium in the fresh curd. Solution: Ensure proper calcium chloride use from the beginning, as texture problems that develop during aging cannot be corrected later.

Storage and Safety Considerations
Proper storage of calcium chloride ensures it remains effective and safe to use over time.
Calcium chloride is hygroscopic, meaning it absorbs moisture from the air. Store it in an airtight container in a cool, dry place away from direct sunlight. If you purchase calcium chloride solution, keep it tightly capped when not in use. Properly stored, calcium chloride has a very long shelf life—several years for powder forms and at least a year for solutions.
Always use food-grade calcium chloride for cheese making. Other grades intended for industrial use, de-icing, or other applications may contain impurities unsuitable for food. Purchase your calcium chloride from reputable suppliers of cheese-making or food ingredients.
When handling calcium chloride, basic kitchen hygiene applies. It’s not hazardous but can be mildly irritating to skin in concentrated form. Wash your hands after handling, and avoid getting it in your eyes. Keep it out of reach of children and pets, as you would with any kitchen ingredient.
Beyond Cheese: Other Uses of Calcium Chloride
While cheese making is a primary food application, calcium chloride has other culinary uses worth noting. It’s used in canning and pickling to maintain the crispness of vegetables, particularly cucumbers. The calcium strengthens pectin in plant cell walls, preventing softening during processing. It’s also used in molecular gastronomy for spherification techniques, in brewing to adjust water chemistry, and in tofu making as a coagulant.
Understanding these broader applications illustrates that calcium chloride is a versatile food ingredient with a well-established safety profile across many culinary applications.
Environmental and Sustainability Aspects
Calcium chloride is generally considered environmentally benign in food applications. It’s a naturally occurring mineral compound and does not persist as a pollutant. The small amounts used in cheese making end up primarily in whey, which is often further processed into animal feed or other products, or is properly treated in wastewater systems where calcium and chloride pose no environmental concerns.
From a sustainability perspective, calcium chloride actually contributes positively by improving cheese yields, meaning more efficient use of milk resources. Better yields translate to less milk needed per unit of cheese produced, reducing the overall environmental footprint of cheese production.
Conclusion: Empowering Your Cheese-Making Journey
Calcium chloride represents one of those seemingly small details that makes a substantial difference in outcome. For anyone working with commercially available pasteurized milk—which describes most cheese makers globally—understanding and using calcium chloride appropriately can be the difference between frustrating results and consistent success.
This modest ingredient restores a natural balance disrupted by necessary safety processing, enabling the creation of quality cheeses with proper texture, good yield, and predictable results. It democratizes cheese making, allowing home enthusiasts to achieve results that would otherwise require access to raw milk or specialized equipment.
Whether you’re just beginning your cheese-making journey or looking to improve your existing practice, incorporating calcium chloride with proper understanding and technique will serve you well. The investment is minimal—both financially and in terms of effort—while the improvements in your cheese-making success can be remarkable.
As you continue exploring the fascinating world of cheese making, remember that success comes from understanding not just what to do, but why. Calcium chloride exemplifies this principle perfectly: a simple addition with profound effects, rooted in the chemistry of milk proteins and the transformative process that turns liquid milk into one of humanity’s most beloved foods.

