Crystallography represents one of nature’s most fascinating phenomena—the formation of perfectly ordered geometric structures at the atomic level. From the intricate patterns of snowflakes to the brilliant facets of gemstones, crystalline structures surround us in countless forms. This comprehensive guide explores the fundamental principles of crystallography, focusing on three primary crystal systems that govern how atoms arrange themselves in solid materials. Whether you’re a student, hobbyist, educator, or simply curious about the natural world, understanding these basic structures opens a window into the hidden architecture that shapes the materials we encounter every day.

What is Crystallography?
Crystallography is the scientific study of crystals and their atomic structures. At its core, a crystal is a solid material whose constituent atoms, molecules, or ions are arranged in an orderly repeating pattern extending in all three spatial dimensions. This internal order gives crystals their characteristic external shapes and distinctive physical properties.
The significance of crystallography extends far beyond academic interest. This field has profound implications across multiple domains:
- Scientific Understanding: Crystallography provides fundamental insights into material properties, helping scientists predict how substances will behave under different conditions, how they’ll interact with light, electricity, and heat, and why they possess specific mechanical characteristics.
- Technological Innovation: Modern technology depends heavily on crystallographic knowledge. From semiconductor manufacturing to pharmaceutical development, understanding crystal structures enables engineers and researchers to design materials with precisely tailored properties. Solar cells, computer chips, LED displays, and fiber optic cables all rely on materials whose crystalline structure determines their functionality.
- Medical Advancement: In pharmaceutical research, crystallography plays a crucial role in drug development. Understanding how drug molecules crystallize affects their stability, solubility, and bioavailability—factors that directly impact therapeutic effectiveness.
- Everyday Materials: Crystals aren’t confined to laboratories or geology collections. Table salt, sugar, ice, metals, minerals, and countless other substances we interact with daily have crystalline structures that determine their characteristics.
The study of crystallography combines principles from physics, chemistry, mathematics, and materials science, making it an inherently interdisciplinary field with applications that touch nearly every aspect of modern life.
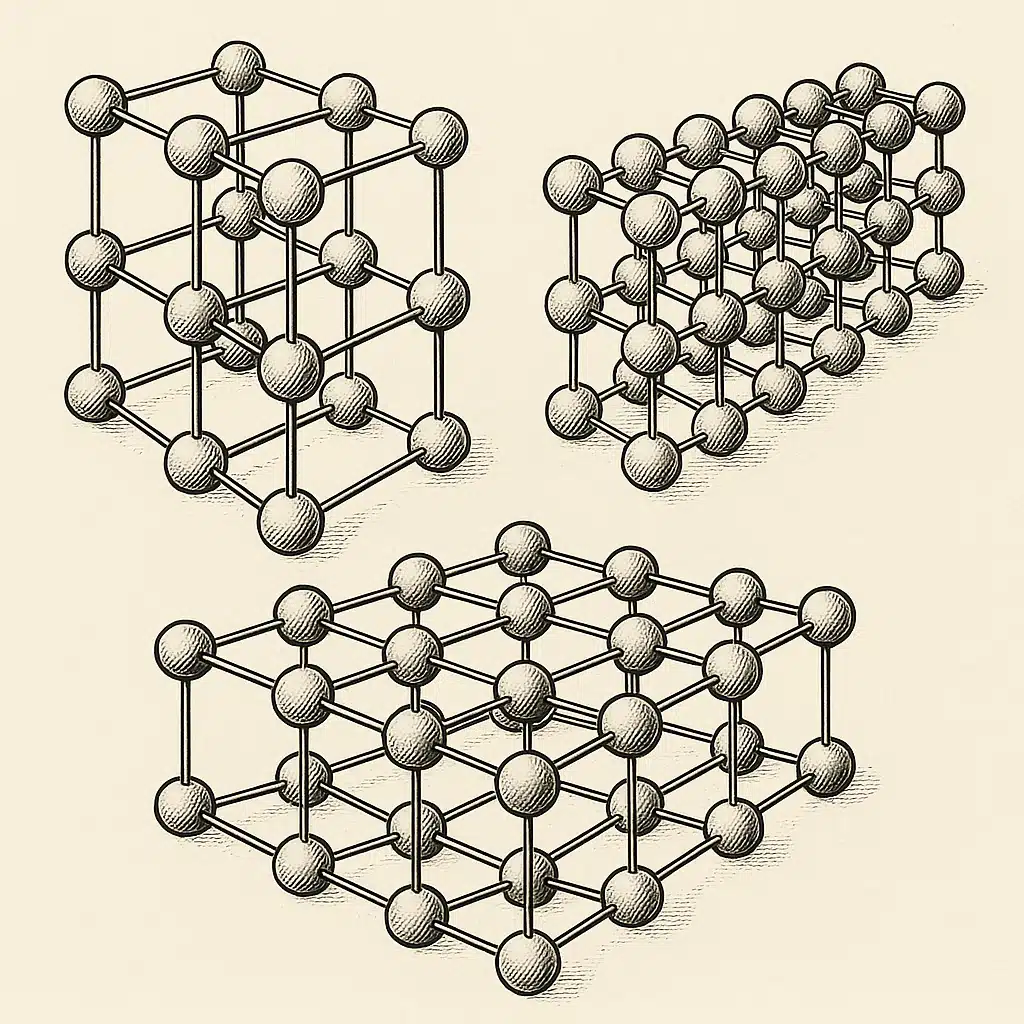
The Crystal Systems: Understanding Atomic Architecture
Scientists have identified seven crystal systems based on the geometric relationships between crystal axes and angles. These systems describe how the fundamental building blocks—called unit cells—repeat throughout space to create macroscopic crystals. In this guide, we’ll focus on three of the most commonly encountered systems: cubic, hexagonal, and monoclinic. Each possesses distinctive characteristics that influence the properties of materials that crystallize in these forms.
The Cubic Crystal System: Maximum Symmetry
The cubic (or isometric) crystal system represents the highest degree of symmetry found in crystalline materials. In this system, the unit cell takes the form of a cube—all three axes are equal in length and intersect at right angles (90 degrees). This high symmetry gives cubic crystals their characteristic appearance and unique properties.
Defining Characteristics:
- Three axes of equal length (a = b = c)
- All axes perpendicular to each other (α = β = γ = 90°)
- High rotational symmetry
- Often forms cube-shaped or octahedral crystals
Common Examples:
- Halite (Sodium Chloride): Common table salt crystallizes in the cubic system, forming characteristic cube-shaped crystals that can be observed with simple magnification.
- Diamond: Perhaps the most famous cubic crystal, diamond’s structure contributes to its exceptional hardness and optical properties.
- Pyrite: Often called “fool’s gold,” this mineral forms striking cubic and pyritohedral crystals.
- Many Metals: Iron, copper, aluminum, gold, and silver all crystallize in cubic forms, which influences their mechanical and electrical properties.
Practical Significance: The cubic structure’s high symmetry often results in isotropic properties—meaning the material behaves similarly regardless of directional orientation. This makes cubic crystals particularly valuable in engineering applications where consistent performance is essential. The electronics industry relies heavily on cubic semiconductors, while metallurgy depends on understanding how cubic metal structures deform and strengthen.

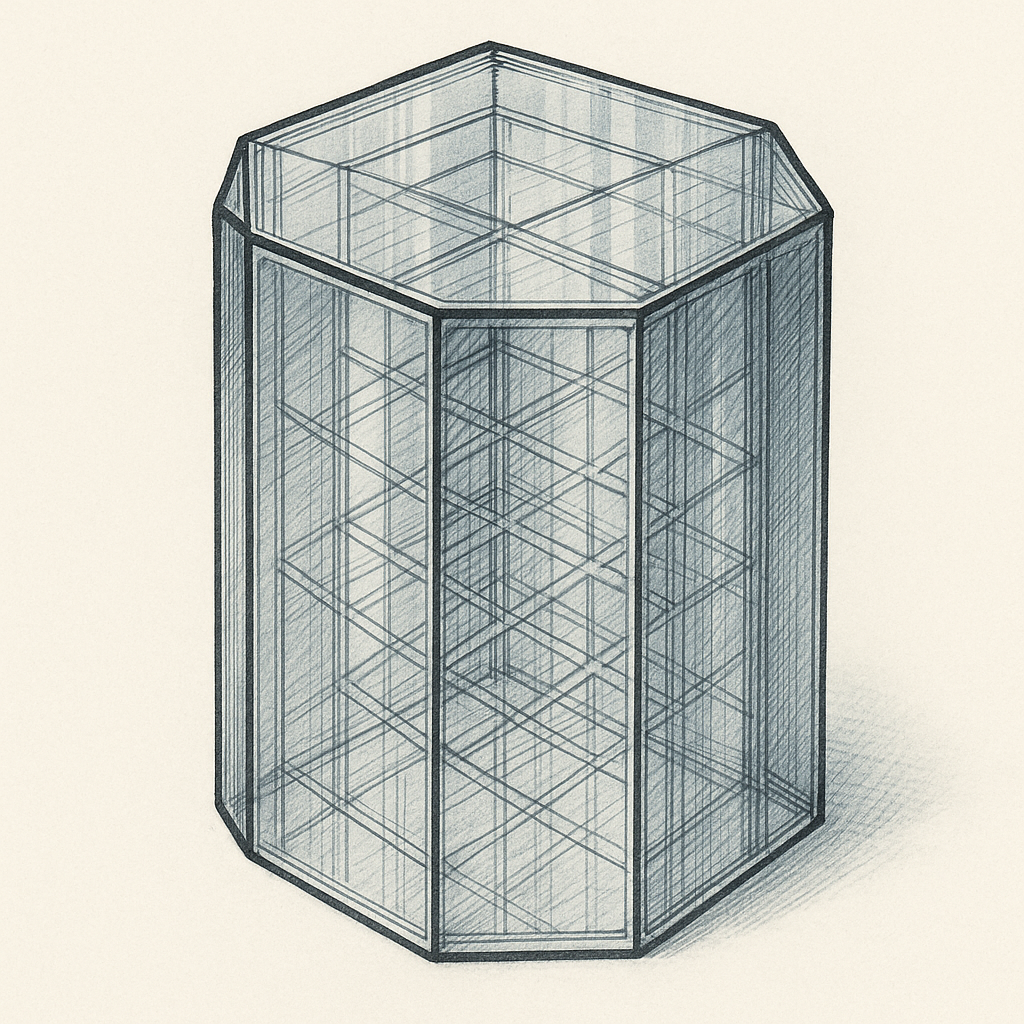
The Hexagonal Crystal System: Six-Fold Symmetry
The hexagonal crystal system is characterized by its distinctive six-fold rotational symmetry. Imagine looking down at a honeycomb—that hexagonal pattern reflects the same symmetry principle governing this crystal system. Hexagonal crystals have four axes: three equal-length axes arranged at 120-degree angles in a horizontal plane, plus one vertical axis of different length perpendicular to the others.
Defining Characteristics:
- Four crystallographic axes (three equal horizontal axes plus one vertical axis)
- The three horizontal axes intersect at 120-degree angles
- The vertical axis is perpendicular to the horizontal plane
- Six-fold rotational symmetry
- Often forms prismatic or pyramidal shapes
Common Examples:
- Quartz: One of Earth’s most abundant minerals, quartz crystals display beautiful hexagonal prisms topped with pyramidal terminations. Their piezoelectric properties (generating electric charge under mechanical stress) make them invaluable in electronic devices.
- Graphite: The same element as diamond (carbon) but arranged in hexagonal sheets that slide easily over each other, creating graphite’s characteristic slippery feel and making it ideal for lubricants and pencil leads.
- Ice: Water’s solid form crystallizes in the hexagonal system, which explains why snowflakes display six-fold symmetry.
- Beryl: This mineral family includes the gemstones emerald and aquamarine, both of which form hexagonal crystals.
- Zinc and Magnesium: These industrially important metals crystallize in hexagonal structures.
Practical Significance: Hexagonal crystals often exhibit anisotropic properties—meaning their characteristics vary depending on direction. Graphite, for example, conducts electricity well within its hexagonal sheets but poorly between layers. This directional dependency is exploited in numerous applications. The timing precision of quartz watches depends on hexagonal quartz crystals, while advanced composites incorporate hexagonal materials to achieve specific strength profiles. In nature, the hexagonal structure of ice influences everything from cloud formation to glacier dynamics.
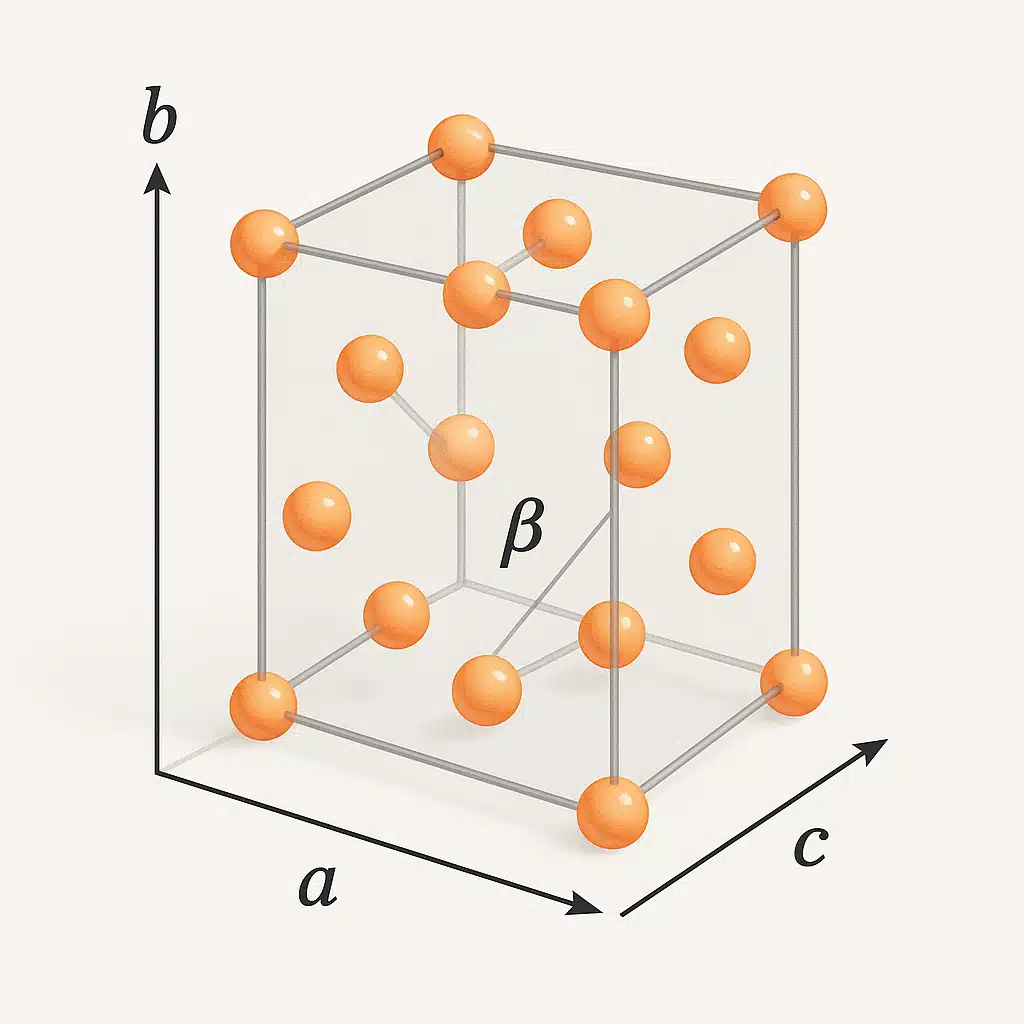
The Monoclinic Crystal System: Asymmetric Complexity
The monoclinic crystal system represents a lower degree of symmetry compared to cubic or hexagonal structures. The term “monoclinic” means “one incline,” referring to the fact that one of the three crystallographic axes is tilted relative to the others. This asymmetry creates distinctive crystal shapes and influences material properties in important ways.
Defining Characteristics:
- Three axes of unequal length (a ≠ b ≠ c)
- Two axes intersect at right angles (90 degrees)
- The third axis is inclined, creating one oblique angle (β ≠ 90°)
- Lower symmetry than cubic or hexagonal systems
- Often forms prismatic or tabular crystals with inclined faces
Common Examples:
- Gypsum: This widespread mineral forms monoclinic crystals and is the primary component of drywall/plasterboard used in construction worldwide. Its various forms include selenite (clear crystals), satin spar (fibrous), and alabaster (massive).
- Orthoclase Feldspar: A major rock-forming mineral found in granite and many other igneous rocks, orthoclase crystallizes in the monoclinic system.
- Mica: This family of minerals, known for splitting into thin flexible sheets, crystallizes in monoclinic forms.
- Azurite and Malachite: These vivid blue and green copper minerals both form monoclinic crystals.
- Some Polymers: Certain synthetic materials, including some plastics, exhibit monoclinic crystallinity.
Practical Significance: The lower symmetry of monoclinic crystals results in more complex physical properties. These materials often show pronounced anisotropy—their optical, mechanical, thermal, and electrical characteristics vary significantly depending on crystallographic direction. In geological contexts, monoclinic minerals provide important information about the conditions under which rocks formed. In materials science, understanding monoclinic structures helps researchers develop specialized materials for applications requiring specific directional properties. The pharmaceutical industry pays particular attention to monoclinic forms of drug compounds, as the crystal structure directly affects how medications dissolve and are absorbed in the body.
Real-World Applications of Crystal Structure Knowledge
Understanding crystal structures transcends academic interest—it provides practical knowledge that drives innovation across numerous fields and impacts daily life in tangible ways.
Medicine and Pharmaceuticals
In pharmaceutical development, the crystalline form of a drug substance can dramatically affect its performance. Different crystal structures of the same chemical compound (called polymorphs) can have vastly different solubility rates, stability profiles, and bioavailability. Researchers use crystallographic techniques to identify the most effective form of a medication, ensuring optimal therapeutic outcomes. The field of crystal engineering focuses specifically on designing drug crystals with desired properties, leading to more effective medications with better storage stability and improved patient outcomes.
Electronics and Technology
Modern electronics depend fundamentally on crystalline materials. Silicon, the foundation of computer chips and solar cells, crystallizes in a cubic (diamond) structure. The precise arrangement of atoms in this crystal lattice allows engineers to control electrical properties with extraordinary precision, creating transistors, diodes, and other semiconductor devices. Liquid crystal displays (LCDs) in screens and monitors use materials that exhibit partial ordering between crystalline solids and liquid states. LED technology relies on crystalline semiconductors that emit light when electrically activated. Even wireless communication depends on crystalline quartz oscillators that provide the timing precision necessary for digital signals.
Materials Engineering
Understanding how metals crystallize allows metallurgists to create alloys with specific properties. Heat treatments that alter crystal structure can make metals harder or more ductile. The development of superalloys for jet engines, structural materials for buildings and bridges, and specialized materials for extreme environments all depend on crystallographic knowledge. Advanced ceramics, composite materials, and nanomaterials are designed with crystal structure considerations at their foundation.
Environmental Science
Crystal structures play important roles in environmental processes. The formation of ice crystals affects cloud formation, precipitation patterns, and climate systems. Mineral crystallization influences soil chemistry, water quality, and geochemical cycles. Understanding how pollutants interact with crystalline minerals helps environmental scientists develop remediation strategies for contaminated sites.
Geology and Earth Sciences
Geologists use crystal structures to understand Earth’s history and processes. The minerals that crystallize from magma reveal information about temperature, pressure, and chemical conditions deep within the planet. Metamorphic changes in crystal structure record the forces that shaped mountain ranges. Even the study of extraterrestrial samples relies on crystallographic analysis to understand the formation of other planets and celestial bodies.

Common Questions About Crystallography
As with any scientific field, crystallography comes with concepts that can seem confusing or counterintuitive. Let’s address some frequently asked questions and clarify common misconceptions.
Are all crystals transparent or gem-like in appearance?
No. While many people associate crystals with transparent gemstones, crystalline materials come in every appearance imaginable. Metals are crystalline but opaque. Graphite is crystalline but looks nothing like a traditional “crystal.” Many crystalline materials appear as powders, masses, or aggregates rather than individual well-formed crystals. The internal atomic order—not external appearance—defines whether something is crystalline.
Do all crystals of the same substance look identical?
Not necessarily. While crystals of the same mineral will share the same internal structure and crystal system, their external appearance can vary significantly based on growth conditions. Factors like temperature, pressure, available space, chemical environment, and growth rate influence crystal shape and size. A quartz crystal formed in a confined space looks different from one that grew freely, even though both have the same hexagonal internal structure.
Is glass a crystal?
No. Despite its transparent, solid appearance, glass is amorphous—meaning its atoms lack the ordered, repeating arrangement that defines crystals. Glass forms when molten material cools too quickly for crystals to organize, resulting in a “frozen liquid” structure. This distinction explains why glass breaks differently than crystalline materials and why its properties are more uniform in all directions.
Can crystal structure change?
Yes. Many substances can crystallize in different forms depending on conditions—a phenomenon called polymorphism. Carbon provides a dramatic example: under normal conditions it forms graphite (hexagonal), but under extreme pressure it becomes diamond (cubic). Temperature and pressure changes can cause crystal structures to transform, which is why materials scientists must consider the conditions under which materials will be used.
Do synthetic crystals differ from natural ones?
Chemically and structurally, synthetic crystals can be identical to natural ones. Laboratory-grown diamonds, rubies, and sapphires have the same crystal structure and properties as mined stones. However, synthetic crystals often have fewer imperfections and more uniform composition than natural specimens. For many technological applications, synthetic crystals are actually superior because their properties can be precisely controlled during growth.

Getting Started with Crystallography
If you’re interested in exploring crystallography further, numerous approaches can deepen your understanding, from hands-on experimentation to theoretical study.
Crystal Growing: One of the most accessible ways to observe crystallography firsthand is growing crystals at home or in educational settings. Simple materials like table salt, sugar, alum, or copper sulfate can produce beautiful crystals that demonstrate fundamental principles. Crystal growing kits provide all necessary materials and instructions, making the process straightforward even for beginners. Observing crystals form over days or weeks provides insight into how atomic ordering creates macroscopic structures.
Mineral Collecting: Building a mineral collection allows you to observe diverse crystal forms in nature. Starting with common, inexpensive specimens representing different crystal systems provides a tangible reference library. Many minerals clearly display their characteristic crystal shapes, making them excellent teaching tools.
Educational Resources: Numerous books, online courses, and video tutorials explain crystallography at various levels. From introductory materials suitable for curious beginners to advanced textbooks for serious students, resources are available to match any learning goal. University websites often provide free educational materials, including interactive crystal structure databases and visualization tools.
Visualization Tools: Software programs allow users to manipulate three-dimensional crystal structures, rotating them to view from different angles and exploring how atoms arrange in space. Many of these tools are available free online, providing powerful learning experiences without requiring specialized equipment.
Community Engagement: Mineral clubs, gem and mineral shows, science centers, and online forums connect people interested in crystallography. These communities offer opportunities to learn from experienced collectors and scientists, view rare specimens, and participate in educational programs.
The Beauty of Atomic Order
Crystallography reveals one of nature’s most elegant principles: that simple rules governing atomic interactions can generate structures of remarkable complexity and beauty. The cubic, hexagonal, and monoclinic systems represent just three of the ways atoms can organize in three-dimensional space, yet these structures encompass countless materials that shape our world.
From the snowflakes drifting through the atmosphere to the silicon crystals powering our digital devices, from the minerals forming mountain ranges to the pharmaceutical crystals healing disease, crystalline structures surround us. Each crystal—whether grown in a laboratory, formed deep within the Earth, or precipitated from solution—reflects the same fundamental geometric principles.
Understanding crystallography provides more than scientific knowledge. It offers a new perspective on the material world, revealing hidden order within seemingly ordinary substances. It connects disciplines from physics to biology, from engineering to art. And it demonstrates how fundamental scientific principles manifest in practical applications that improve human life.
Whether you’re a student beginning to explore materials science, a hobbyist fascinated by minerals, an educator looking to explain natural phenomena, or simply someone curious about how the world works, crystallography offers endless opportunities for discovery. The next time you encounter a crystal—in jewelry, in technology, in nature, or in everyday objects—you’ll recognize the geometric principles at work, appreciating the atomic architecture that gives each material its unique properties.
The study of crystallography continues to advance, driving innovations in medicine, technology, materials science, and environmental protection. By understanding these fundamental structures, we gain tools to design better materials, develop new technologies, and solve complex challenges. In crystallography, science and beauty converge, revealing nature’s perfect geometry in all its manifestations.

