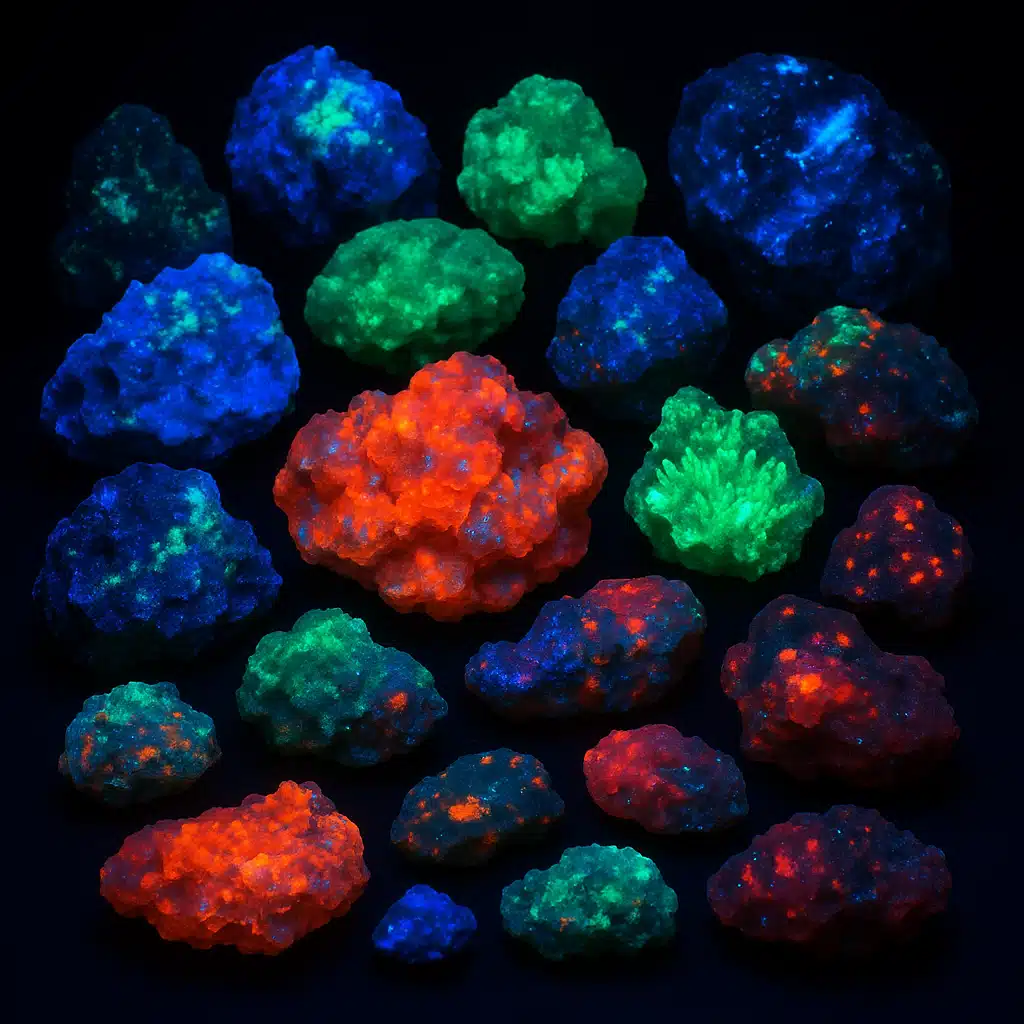
Have you ever wondered why some rocks seem to glow under ultraviolet (UV) light? It’s a fascinating phenomenon that reveals the hidden beauty of the natural world and opens a window into the remarkable properties of minerals. Welcome to the secret world of UV minerals and fluorescence! In this comprehensive guide, we’ll take you on an illuminating journey to understand why certain rocks shine bright when exposed to UV light, explore the science behind this captivating phenomenon, discover which minerals exhibit this property, and learn why it matters for science, education, and collectors worldwide.

What is Fluorescence?
Fluorescence is a natural optical phenomenon that allows certain materials, including rocks and minerals, to absorb electromagnetic radiation at one wavelength and then emit it at a different, typically longer wavelength. When exposed to ultraviolet light—which is invisible to the human eye—fluorescent minerals absorb this high-energy radiation and almost instantly release it as visible light in various colors such as green, blue, red, orange, or yellow.
Think of it like a sophisticated light conversion system: the mineral acts as both a receiver and a transmitter. The UV light provides energy that excites electrons within the mineral’s atomic structure, temporarily pushing them to higher energy states. When these electrons return to their normal state, they release the excess energy as visible light—creating that magical glow that captivates scientists and hobbyists alike.
What makes fluorescence particularly special is that it occurs only while the UV light source is active. The moment you switch off the UV light, most fluorescent minerals stop glowing immediately, distinguishing this phenomenon from phosphorescence, where materials continue to emit light even after the excitation source is removed.
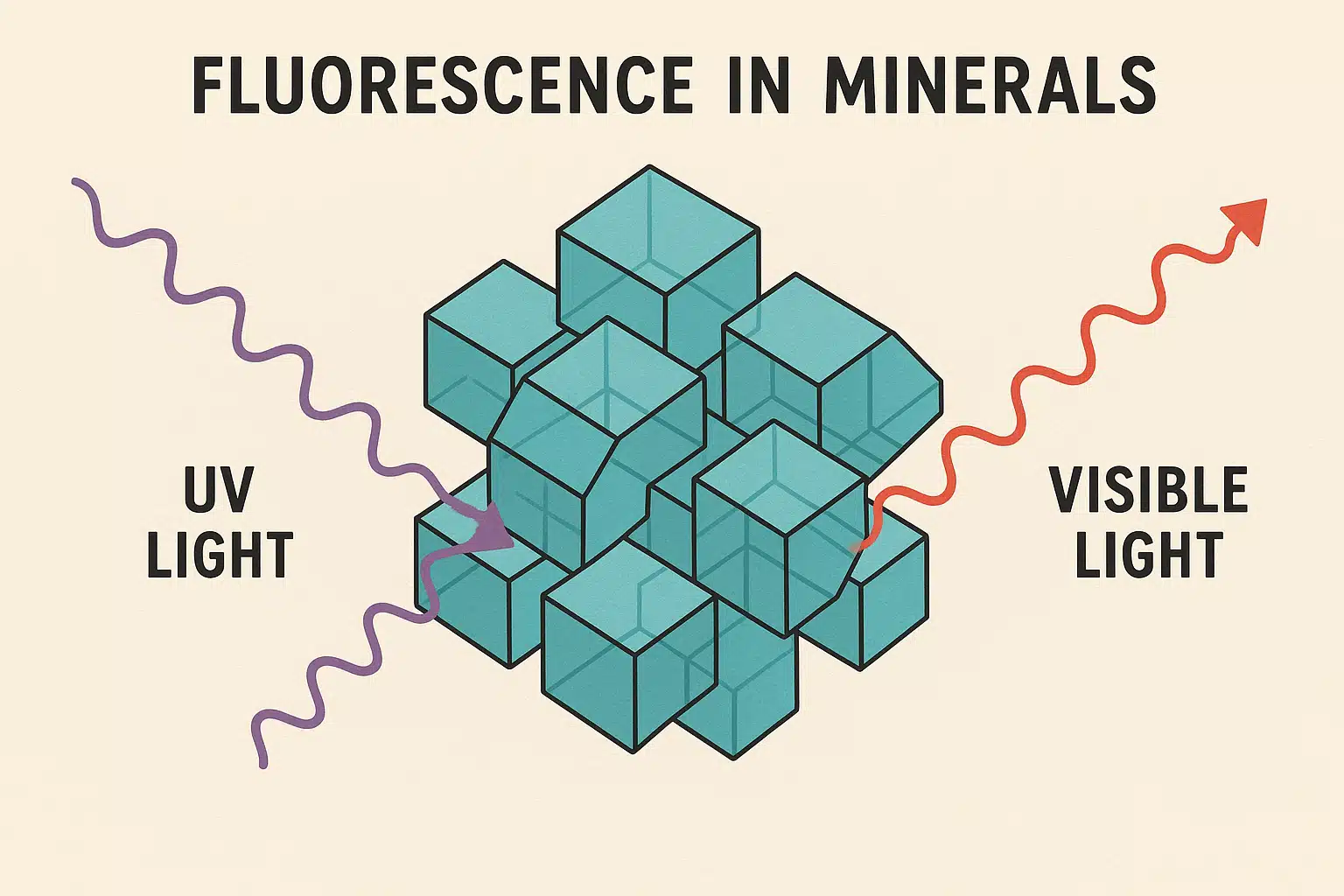
Why Do Some Rocks Glow While Others Don’t?
Not all rocks and minerals exhibit fluorescence, and understanding why requires looking at several key factors that influence this phenomenon:
Mineral Composition
The chemical composition of a mineral is the primary factor determining whether it will fluoresce. Certain elements and compounds have atomic structures that naturally facilitate the absorption and re-emission of light energy. Minerals containing elements such as zinc, calcium, manganese, barium, uranium, or rare earth elements are more likely to display fluorescent properties. The specific arrangement of atoms within the mineral’s crystal lattice also plays a crucial role in how efficiently energy can be absorbed and released.
Activator Elements and Impurities
The presence of trace elements, often called “activators,” can dramatically enhance or even enable fluorescence in minerals that otherwise wouldn’t glow. These impurities—which may be present in concentrations of less than one percent—act as the actual fluorescent centers within the mineral. For example, manganese impurities can cause calcite to fluoresce red or orange, while tungsten can create blue fluorescence in scheelite. Interestingly, certain impurities can also act as “quenchers,” suppressing or completely blocking fluorescence.
UV Light Wavelength
Not all ultraviolet light is created equal. UV radiation is divided into different ranges: longwave UV (315-400 nm), midwave UV (280-315 nm), and shortwave UV (100-280 nm). Different minerals respond to different UV wavelengths. Some minerals fluoresce brilliantly under longwave UV but show no response to shortwave UV, while others display the opposite behavior. Some minerals even change color depending on which UV wavelength is used, revealing multiple fluorescent responses within the same specimen.
Crystal Structure and Defects
The internal structure of a mineral, including any defects, fractures, or irregularities in its crystal lattice, can significantly affect fluorescence. Sometimes these structural imperfections create energy states that enable fluorescence, while in other cases they may reduce or alter the fluorescent response.

Real-Life Examples of Fluorescent Minerals
The world of fluorescent minerals is incredibly diverse, with hundreds of mineral species displaying this remarkable property. Here are some of the most notable and widely recognized fluorescent minerals that captivate collectors and scientists around the globe:
Fluorite
Perhaps the most famous fluorescent mineral, fluorite derives its name from this very property. This calcium fluoride mineral can display a stunning range of fluorescent colors including blue, violet, green, yellow, and even white, depending on the activator elements present and the UV wavelength used. Under longwave UV light, many fluorite specimens glow a brilliant blue or purple. Fluorite’s consistent and vibrant fluorescence makes it a favorite among collectors worldwide, and it’s often used in educational demonstrations and decorative mineral displays.
Calcite
Calcite, one of the most common minerals on Earth, exhibits highly variable fluorescence depending on its manganese content and other trace elements. Pure calcite typically doesn’t fluoresce, but when manganese is present, it can glow brilliant red, orange, pink, or even yellow under UV light. This variability makes calcite specimens particularly interesting to collectors, as similar-looking samples can produce dramatically different fluorescent responses.
Willemite
Willemite, a zinc silicate mineral, is renowned for its intense bright green fluorescence under shortwave UV light, caused by manganese activation. This mineral creates some of the most vivid and striking fluorescent displays in the mineral world. When found alongside other fluorescent minerals like calcite (red) and franklinite (non-fluorescent black), willemite creates stunning multi-colored fluorescent scenes that are highly prized by collectors.
Scheelite
This calcium tungstate mineral typically fluoresces bright blue or white under shortwave UV light. Scheelite’s reliable fluorescence has practical applications in mining operations, where UV lamps are used to locate scheelite ore deposits in the field, making extraction more efficient and cost-effective.
Aragonite
A polymorph of calcite with the same chemical composition but different crystal structure, aragonite often displays variable fluorescence ranging from blue-white to pink, yellow, or green. Its fluorescent properties vary significantly depending on the presence of activator elements and the geological environment in which it formed.
Sodalite
This sodium aluminum silicate mineral typically fluoresces orange or yellow under longwave UV light. Sodalite’s intense fluorescence combined with its attractive blue color in normal light makes it popular for both mineral collections and ornamental applications.
Hyalite Opal
This colorless or white variety of opal can exhibit brilliant green fluorescence under UV light due to uranium content. Hyalite specimens create an otherworldly appearance, especially when the opal forms botryoidal (grape-like) clusters that seem to glow from within.

Common Misunderstandings About Fluorescence
Despite the growing interest in fluorescent minerals, several misconceptions persist about this phenomenon. Understanding these common misunderstandings helps develop a more accurate appreciation of mineral fluorescence:
Not All Rocks Glow Under UV Light
Perhaps the most prevalent misconception is that all or most rocks will fluoresce when exposed to UV light. In reality, only a relatively small percentage of minerals display fluorescence, and among those that do, the intensity and visibility of the fluorescence can vary dramatically. Many common rocks and minerals show no fluorescent response whatsoever, regardless of the UV wavelength used.
Fluorescence vs. Phosphorescence
Many people confuse fluorescence with phosphorescence, but these are distinctly different phenomena. Fluorescence occurs only while the UV light source is active—turn off the light, and the glow disappears almost instantly. Phosphorescence, on the other hand, describes materials that continue to emit light for seconds, minutes, or even hours after the excitation source is removed. While some minerals exhibit both properties, they are separate phenomena with different mechanisms at the atomic level.
Fluorescence Doesn’t Indicate Radioactivity
Some people assume that fluorescent minerals are radioactive or dangerous. While certain fluorescent minerals do contain radioactive elements like uranium (such as autunite or hyalite opal), the vast majority of fluorescent minerals are completely non-radioactive and perfectly safe to handle. Fluorescence is an optical property, not an indicator of radiation.
Fluorescence Is Not Always Predictable
Even among the same mineral species from the same location, fluorescence can vary significantly or be absent entirely. Two calcite specimens that look identical in normal light may show completely different fluorescent responses due to variations in trace element content. This unpredictability is part of what makes fluorescent mineral collecting so exciting.
UV Light Quality Matters Significantly
Not all UV lights produce the same results. Consumer-grade “black lights” typically produce only longwave UV radiation, which means they’ll miss fluorescence that only responds to shortwave or midwave UV. Professional mineral collectors and researchers use specialized UV lamps that can produce different wavelengths to fully characterize a specimen’s fluorescent properties.

Why Understanding Fluorescence Is Important
The study and application of mineral fluorescence extends far beyond simple curiosity or aesthetic appreciation. Understanding fluorescence has practical importance across multiple fields and industries worldwide:
Mineralogy and Geology
Fluorescence serves as a valuable diagnostic tool for geologists and mineralogists. The fluorescent response of a mineral—including the color, intensity, and which UV wavelength produces the response—provides important clues about its chemical composition, trace element content, and formation environment. This information helps scientists identify minerals, distinguish between similar-looking species, and understand the geological processes that created them. Fluorescence analysis is now a standard technique in professional mineralogical laboratories worldwide.
Mining and Resource Extraction
Fluorescence has direct practical applications in the mining industry. Portable UV lamps are used in the field to locate ore deposits of valuable minerals like scheelite (tungsten ore), which fluoresces bright blue, making it visible even in low-grade deposits that might otherwise be missed. This technique, called “lamp prospecting,” has proven effective in improving exploration efficiency and reducing costs. Fluorescence is also used in ore sorting operations, where automated systems can separate fluorescent minerals from waste rock, improving processing efficiency.
Education and Scientific Outreach
Fluorescent minerals provide powerful hands-on learning experiences in educational settings from elementary schools to universities. The dramatic visual transformation that occurs when UV light illuminates a seemingly ordinary rock captures attention and sparks curiosity about earth sciences, chemistry, and physics. Science museums worldwide feature fluorescent mineral displays that consistently rank among their most popular exhibits, introducing countless visitors to the hidden wonders of the mineral kingdom.
Authentication and Gemology
In the gemstone industry, fluorescence testing is a standard procedure for identifying and authenticating gems. Different gemstone materials show characteristic fluorescent responses that help gemologists distinguish natural stones from synthetic ones, identify treatments, and detect imitations. For example, natural diamonds often show distinctive blue fluorescence under UV light, while certain treatments or synthetic diamonds may show different responses or no fluorescence at all.
Scientific Research
Fluorescence studies contribute to our understanding of crystal chemistry, solid-state physics, and geological processes. Research into mineral fluorescence has led to discoveries about how trace elements are incorporated into crystal structures, how minerals form in different environments, and how energy transfer occurs at the atomic level. This fundamental research has applications beyond geology, influencing fields like materials science and optoelectronics.
Hobby and Collecting Community
Fluorescent mineral collecting has developed into a specialized hobby with a dedicated global community. Collectors seek rare fluorescent specimens, attend specialized shows and exhibitions, and share their discoveries through online communities and publications. This vibrant hobby promotes appreciation for earth sciences and contributes to citizen science through documented observations and discoveries of fluorescence in new localities or mineral species.
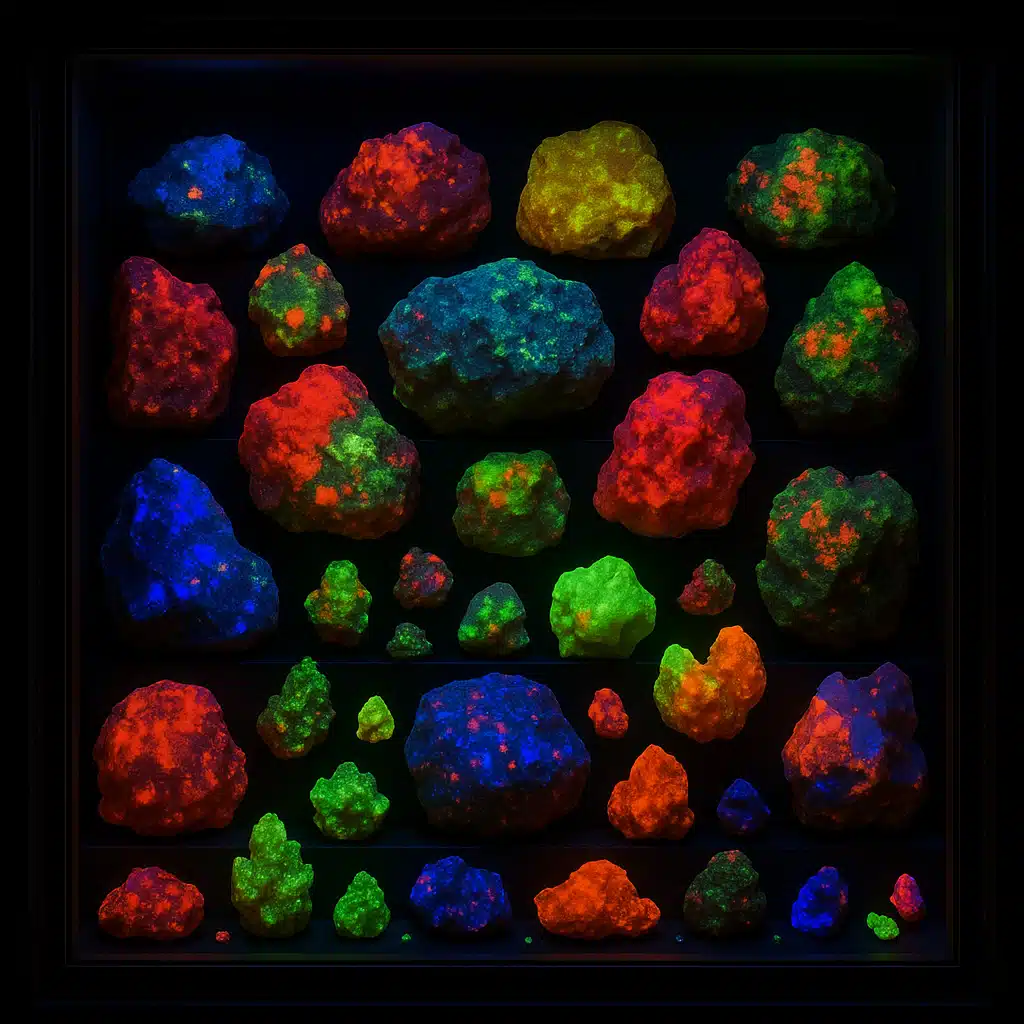
Getting Started With Fluorescent Minerals
If you’re interested in exploring the fascinating world of fluorescent minerals, getting started is more accessible than you might think. Here are some practical tips for beginning your journey into this captivating aspect of earth sciences:
Acquire a Quality UV Light
The foundation of fluorescent mineral exploration is a good UV light source. Consider starting with a dual-wavelength lamp that provides both longwave (365nm) and shortwave (254nm) UV light, as different minerals respond to different wavelengths. Portable, battery-powered UV flashlights are convenient for field work and initial exploration, while more serious collectors invest in higher-powered lamps for detailed examination and photography.
Start With Known Fluorescent Specimens
Begin your collection with minerals known for reliable, vivid fluorescence such as fluorite, calcite, sodalite, or willemite. These provide immediate gratification and help you understand what to look for. Many mineral dealers specifically label specimens that show fluorescence, making it easier for beginners to build a foundational collection.
Practice Safety Precautions
UV radiation, particularly shortwave UV, can be harmful to eyes and skin with prolonged exposure. Always use appropriate UV-blocking safety glasses when examining minerals with shortwave UV light, avoid looking directly at the UV lamp, and limit exposure time. Use UV light in well-ventilated areas and keep it away from children without supervision.
Create a Proper Viewing Environment
Fluorescence is best observed in complete darkness or very low ambient light conditions. Set up a viewing area where you can control lighting, such as a darkened room or a viewing box. This allows you to fully appreciate subtle fluorescence that might be invisible in bright conditions.
Document Your Discoveries
Keep records of your fluorescent specimens, noting the mineral name, locality, fluorescent colors under different UV wavelengths, and intensity. Photography of fluorescent minerals presents unique challenges but can produce stunning images. Consider joining online communities where collectors share techniques, discoveries, and experiences.
The Future of Fluorescent Mineral Studies
Research into mineral fluorescence continues to advance our understanding of these phenomena. Modern analytical techniques, including spectroscopy and advanced imaging, are revealing new details about the mechanisms behind fluorescence and discovering previously unknown fluorescent minerals. As technology improves, portable analytical equipment becomes more accessible, allowing field identification and analysis that was once possible only in laboratories.
The growing interest in fluorescent minerals has also spurred increased exploration and documentation of fluorescent occurrences worldwide. Collectors and researchers are discovering that many previously unknown localities contain fluorescent minerals, expanding our knowledge of their distribution and variety. This collaborative effort between amateur collectors and professional scientists continues to contribute valuable data to the mineralogical community.
Conclusion
The secret world of UV minerals offers a captivating window into the hidden properties of the natural world. Fluorescence reveals that there is literally more to rocks and minerals than meets the eye—a whole spectrum of colors and behaviors invisible under normal lighting conditions. By understanding the science behind fluorescence, recognizing which minerals exhibit this property, and appreciating its practical applications, we gain deeper insight into both the beauty and utility of the mineral kingdom.
Whether you’re a professional geologist, an educator, a collector, or simply someone curious about the natural world, fluorescent minerals offer endless opportunities for discovery and wonder. From the brilliant blue glow of fluorite to the intense green luminescence of willemite, these glowing rocks remind us that nature still holds many secrets waiting to be illuminated. The study of fluorescent minerals bridges art and science, combining aesthetic appreciation with rigorous scientific investigation, making it accessible and rewarding for anyone willing to look beneath the surface.
As you explore this fascinating field, remember that every fluorescent specimen has a unique story written in the light it emits—a story of geological processes, chemical composition, and the fundamental behavior of matter and energy. So grab a UV light, dim the room, and prepare to see the mineral world in an entirely new light. The glowing rocks are waiting to share their secrets with you.